 
This is the origin of the electrostatic repulsion between two membrane surfaces in an aqueous ionic medium, as distinct from air. A net negatively-charged membrane in an electrolyte is surrounded by a diffuse layer of counterions and is therefore electrically neutral however, bringing two such membranes together would squeeze the diffuse counterion layers which then resist with an osmotic pressure. It partly determines the separation between stacked thylakoids, recently estimated as 3.2 nm 13.Įlectrostatic repulsion, caused by the net negative charge on the outer surface of thylakoid membranes, is a major factor opposing granal stacking 5, 14, 15, 16, 17, 18. Hydration repulsion is very short-ranged, originating from the displacement of structured water molecules around charged groups on membranes surfaces 12. Granal stacking should depend on the net sum of (1) hydration repulsion, (2) electrostatic repulsion, (3) van der Waals attraction and (4) entropy-driven attraction. 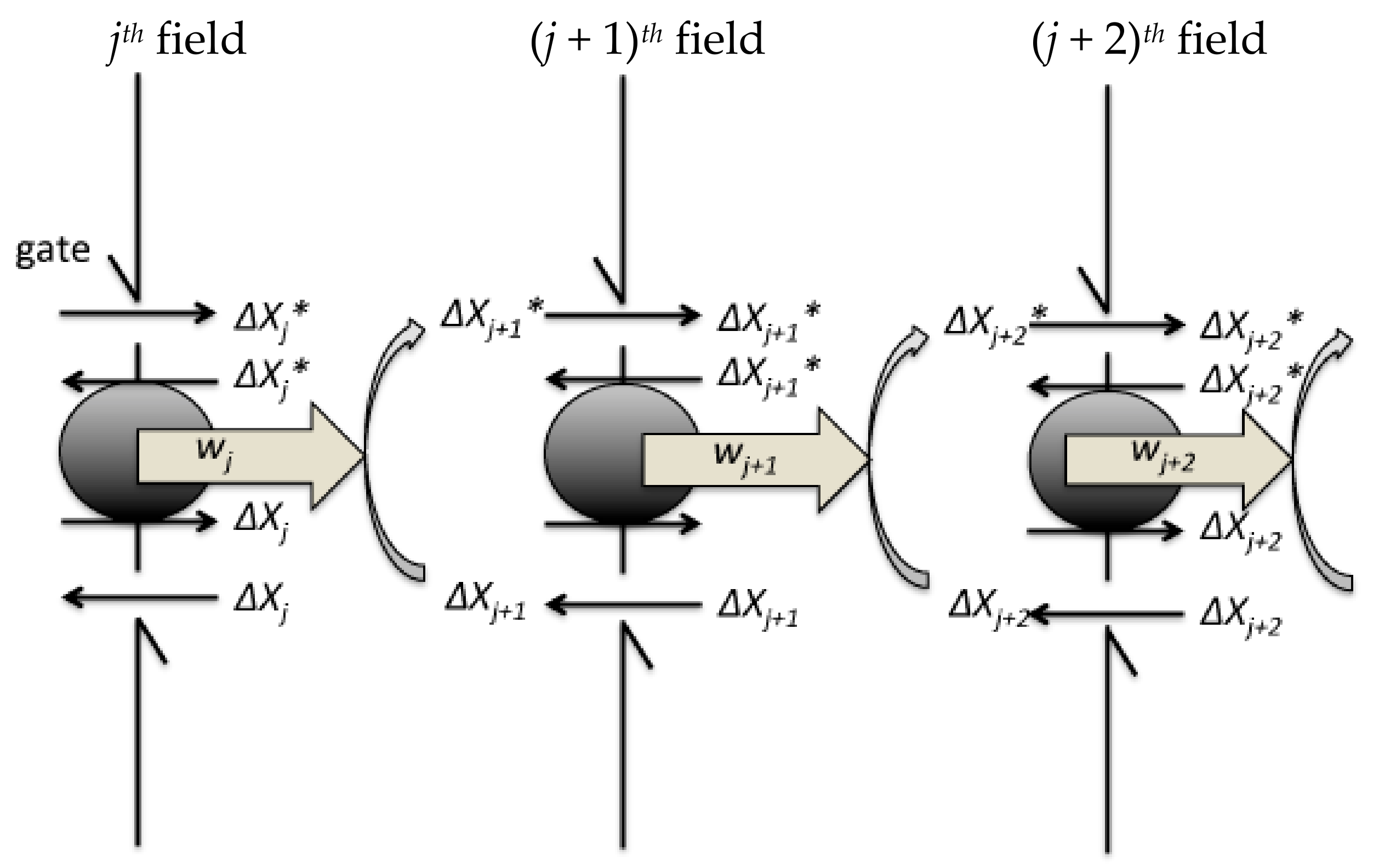 
Here we monitored Mg 2+-induced stacking of spinach photosynthetic membranes to address the question of what thermodynamic attractive force drives granal stacking. Advantages of granal stacking include: (1) an extremely large membrane surface-to-volume ratio to accommodate a high density of chlorophyll (Chl) to promote light-harvesting 5 (2) a spatial separation of the two photosystems 6 which limits excessive spillover of excitation energy from Photosystem II (PS II, responsible for oxygen evolution) to PS I 1, 2, 7 (3) enhancement of non-cyclic photophosphorylation to increase the “energy currency” 8 (4) regulation of non-photochemical dissipation for protection against excess light 9 (5) delay of premature degradation of D1 protein in the PS II reaction centre at sustained high irradiance 10 (6) regulation of linear versus cyclic electron transport 11 and (7) a potential increase in photosynthetic capacity for a given chloroplast composition 4. Stacking of photosynthetic membranes (thylakoids) to form grana interconnected by non-stacked thylakoids, bathed in an aqueous stroma, is ubiquitous in plants, being required for fine-tuning (a) photosynthesis, (b) photoprotection and (c) acclimation to environments 1, 2, 3, 4. Possible mechanisms for the entropy increase include: (i) the attraction between discrete oppositely-charged areas, releasing counterions (ii) the release of loosely-bound water molecules from the inter-membrane gap (iii) the increased orientational freedom of previously-aligned water dipoles and (iv) the lateral rearrangement of membrane components. This first direct experimental evidence strongly suggests that an entropy increase significantly drives membrane stacking in this ordered biological structure. 
After subtracting a large endothermic interaction of MgCl 2 with membranes, unrelated to stacking, we demonstrate that the enthalpy change (heat change at constant pressure) is zero or marginally positive or negative. Here, for the first time, we apply Isothermal Titration Calorimetry (ITC) to investigate the Mg 2+-induced spontaneous stacking of photosynthetic membranes isolated from spinach leaves. According to the Second Law of Thermodynamics, an overall increase of entropy contributes to the driving force for any physicochemical process, but entropy has seldom been investigated in biological systems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
